Abstract
The rate of oxidation of SO2 was studied in a wine-model system under aerial oxygen saturation conditions to gain further insight into its mechanism of action in wine. When SO2 was studied alone, no significant oxidation was observed unless iron and copper were introduced. When these metals were added a slow oxidation was observed and bound SO2 also increased to a small but significant extent. These results are consistent with a radical chain mechanism initiated by metal catalysis, in which powerful oxidizing radicals, capable of oxidizing ethanol to acetaldehyde, are produced. This increase in bound SO2 is prevented by 4-methylcatechol (4-MeC) in keeping with the known ability of polyphenols to scavenge these intermediate radicals and thus to inhibit SO2 autoxidation, which consequently should not occur in wine. When 4-MeC was introduced at a concentration that simulated the reducing capacity of red wine, again no significant SO2 oxidation was observed without addition of iron and copper. Had the catechol been oxidized, hydrogen peroxide would have been generated and reacted with the SO2. In the presence of both metals the rate of SO2 oxidation was markedly increased compared to SO2 alone and then was dependent on the concentration of the catechol. These results demonstrate the crucial importance of metals in allowing polyphenol oxidation and that the rate of SO2 consumption is dependent on the rate of catechol oxidation. When iron and copper were added separately, only a modest increase in rate of catechol oxidation resulted. However, when combined, marked synergism was observed and the rate then became very sensitive to copper concentration. It is proposed that copper, by interacting with oxygen, facilitates redox cycling of iron. Exposure of a red wine to the conditions used in this study produced similar results regarding SO2 oxidation to those observed.
Sulfur dioxide has become virtually indispensable in winemaking. This apparently simple substance combines three important beneficial properties: antimicrobial and antioxidant activity and the ability to add to carbonyl compounds to form nonvolatile bisulfite adducts, so preventing their undesirable sensory properties. After long usage and extensive study, SO2 is accepted as having low toxicity. However, it has become apparent that it can induce allergic reactions (Ribéreau-Gayon et al. 2000). For this reason and the general public’s increasing suspicion of any food or drink additive, the trend is to minimize usage in wine. A full understanding of its various mechanisms of action is therefore essential for its appropriate usage and indeed, perhaps one day, to eliminate its use entirely with the discovery of a superior alternative.
Some researchers are of the opinion that SO2 reacts with oxygen directly and so protects polyphenols and other wine constituents from oxidation (Ribéreau-Gayon et al. 2000, Clarke and Bakker 2004). However, studies in model wines, with respect to ethanol content and pH but where polyphenols are excluded, have shown that the reaction of SO2 with oxygen is extremely slow when compared with the uptake of oxygen by wine itself. Consequently, it is concluded by others that the prime function of SO2 is to scavenge hydrogen peroxide produced by the oxidation of polyphenols (Boulton et al. 1996). These very different views must result in some confusion in those who are attempting to understand how SO2 functions as an antioxidant in wine.
The interaction of SO2 with oxygen has been studied for over one hundred years, mainly because of its importance in the desulfurization of flue gases and production of acid rain in the atmosphere, where pH is ~4; not too distant from that of wine (Brandt and van Eldik 1995). In aqueous systems SO2 combines with water to produce sulfurous acid, which dissociates as shown in reaction 1, such that in wine the bisulfite form (HSO3−) predominates. The reaction with oxygen may be represented by reaction 2, where two moles of bisulfite react simply with one mole of oxygen to produce two moles of sulfate. However, drawn in this manner, it could imply that bisulfite reacts directly with molecular oxygen, which is highly misleading. The actual mechanism by which it is converted to sulfate is quite complex, which has important implications regarding the chemical transformations it can induce in wine. There is a fundamental reason why oxygen cannot interact directly in one step with SO2 or bisulfite and sulfite ions. The outer electrons of the oxygen molecule that are available for forming covalent bonds are in separate molecular orbitals with their spins aligned. In normal conditions, therefore, molecular oxygen is in a triplet ground state; in effect a di-radical. Such a molecule cannot interact directly with species such as polyphenols and SO2, which possess paired electrons with antiparallel spins (singlet state). If this reaction did occur, it would result in new molecular orbitals containing electrons with parallel spins, which would violate Pauli’s exclusion principle. The reaction could only proceed with spin inversion to the singlet state, which would require a substantial energy input. Without such input, reactions with oxygen proceed in one-electron steps with the formation of free radicals (Danilewicz 2003).


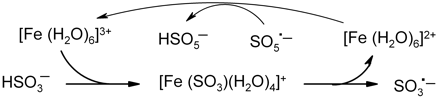
The autoxidation of SO2 is catalyzed by transition metals such as iron, copper, and manganese. An uncatalyzed reaction has been described (Connick et al. 1995), but its existence is uncertain as even traces of metals, which can be inadvertently introduced, have a marked effect on reaction rate (Brandt and van Eldik 1995). In acid conditions bisulfite forms metal-sulfito complexes, as represented in scheme 1⇓, using hexa-aquo iron(III) as an example. Inner-sphere electron transfer results in the production of a sulfite radical (SO3·−) with the concomitant reduction of iron to the ferrous state. Iron(III) forms complexes with tartaric and other α-hydroxy acids (Yokio et al. 1994), and thus in wine different mixtures of sulfito complexes will result, but the same net redox process should still operate. Fe(II) is then reoxidized to the ferric state by oxygen-derived species, such as the peroxomonosulfate radical, to restore catalytic function of the metal (i.e., redox cycling).
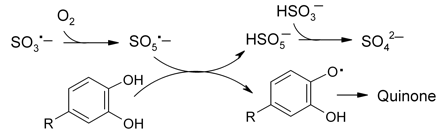
As shown in scheme 2⇓, the sulfite radical reacts rapidly with oxygen to form the peroxomonosulfate radical (SO5·−) (Brandt and van Eldik 1995, Brandt et al. 1994, Connick et al. 1995). This is a strong oxidant, the formal reduction potential (E0) for the SO5·−/HSO5− couple being 1.41 V (Das et al. 1999). The radical is more effective than oxygen in recycling ferrous iron back to the ferric state (scheme 1⇓) and will oxidize bisulfite. The experimental evidence indicates two pathways (A and B) as shown in scheme 2⇓. In path A, the peroxomonosulfate radical oxidizes bisulfite to regenerate the sulfite radical with formation of the peroxomonosulfate ion (HSO5−). This in turn reacts with bisulfite to form disulfate (S2O72−), which rapidly hydrolyzes to sulfate (Connick et al. 1993). In path B the peroxomonosulfate radical oxidizes bisulfite to produce sulfate and the sulfate radical (SO4·−). This radical is a very powerful oxidant (E0 = 2.4 V for the SO4·−/HSO4− couple) of similar oxidizing capability to the hydroxyl radical, and it can therefore oxidize saturated hydroxy compounds. That explains why radical scavengers such as mannitol and ethanol inhibit bisulfite oxidation by intercepting these radicals and so blocking route B (Brandt and van Eldik 1995). The sulfate radical readily oxidizes the bisulfite ion to the bisulfite radical, so completing the cycle with the formation of sulfate (path C). Overall, therefore, as the SO3·− radical enters each cycle, one molecule of oxygen is consumed with a total of two bisulfite ions (via routes A + B) to produce two sulfate ions with the regeneration of one SO3·− radical to continue the chain reaction.
In contrast, the reaction of hydrogen peroxide with SO2 involves neither metals nor free radicals. It is represented as nucleophilic displacement of water from the bisulfite ion (Halperin and Taube 1952, McArdle and Hoffmann 1983, Brandt and van Eldik 1995). However, it is also possible that displacement of water occurs in aquated SO2, as shown in scheme 3 ⇓(Vincent et al. 1998). Whatever the precise mechanism, persulfurous acid is formed, which rearranges to sulfate.
The purpose of this study was to examine the effect of iron and copper on the rate of reaction of SO2 in a simplified wine-model system, both alone and in the presence of a catechol at concentrations that simulate white and red wine conditions. The aim was to provide additional evidence as to the involvement of these metals in the oxidative process and to establish to what extent SO2 can protect, if at all, catechols and other wine constituents from aerial oxidation.
Materials and Methods
Materials.
Water (ISO 3696:1987 grade 3; Fe, Cu < 5 μg/L), iron(II) sulfate heptahydrate, copper(II) sulfate pentahydrate, sodium hydroxide, and (+)-tartaric acid (Fe < 3 μg/g) were AnalaR grade. Ethanol (96% v/v; Fe < 0.8 mg/L) was GPR grade, except when the effect of metals was studied, when AnalaR grade was used (Fe < 0.16 mg/L, Cu < 0.016 mg/L). All materials were BDH branded products (VWR International Ltd., Lutherworth, UK). Potassium metabisulfite, (+)-catechin, 4-methylcatechol, gallic acid, and pyrogallol were obtained from Sigma-Aldrich (Poole, Dorset, UK). The wine studied was Mouton Cadet, appellation Bordeaux Controlée 2001, Baron Philippe de Rothschild SA, Pauillac, France. Alcohol was 12% (v/v); pH 3.6; and free and bound SO2 were <1.5 and 27 mg/L, respectively. The 1H-NMR spectrum of (+)-catechin was obtained in DMSO-d6 using a Mercury-400BB “spect 30” spectrometer (Varian, Palo Alto, CA). pH was measured with a 210 benchmeter (Hanna Instruments, Leighton Buzzard, UK) calibrated at pH 4.00 and 7.00.
Wine model solution.
The model system consisted of 12% (v/v) aqueous ethanol containing tartaric acid with pH adjusted to 3.6. The acid, apart from its function to maintain acidity, is important in coordinating preferentially to ferric ions to reduce the formal reduction potential of the Fe3+/Fe2+ couple so as to facilitate redox cycling (Danilewicz 2003).
(+)-Tartaric acid (8.0 g) was dissolved in water (~800 mL) in a 1-L volumetric flask. Ethanol (125 mL) was added to give a 12% (v/v) final concentration. The required volumes of iron(II) sulfate, and copper(II) sulfate standard solutions (freshly made up) were then added to give the required Fe and Cu concentrations, followed by solid potassium metabisulfite to give a final free SO2 concentration of ~90 mg/L. The pH was increased to 3.60 with 2.5 N sodium hydroxide, also adding water progressively to the mark as the required pH was approached. The titratable acidity of this final model solution was 4.1 g/L expressed as tartaric acid. Initial free SO2 concentrations were determined using 10-mL samples by the potassium iodate/potassium iodide-starch modified Ripper method. Duplicate determinations differed by 1 mg/L or less. The method has the advantage of rapidity but was not thought reliable enough to provide values for bound SO2. The aeration-oxidation method was then used when free and bound SO2 concentrations needed to be determined, as indicated in the text (Ough and Amerine 1988). Typically, when free SO2 was adjusted to ~90 mg/L in freshly made up model solutions, bound SO2 was 4 to 5 mg/L, presumably due to varying levels of acetaldehyde contamination in different batches of ethanol.
Aliquots of 100 mL of the above solutions were placed in 500-mL volumetric flasks, washing in the required amount of polyphenol (gallic acid and (+)-catechin were dissolved completely in ~5 hr with periodic shaking). This afforded ~440-mL air headspace, which provided adequate oxygen exposure, as stirring was found to be unnecessary even at the most rapid rates of SO2 oxidation. When the effect of varying the amounts of iron and copper was studied, it was found equally acceptable and more convenient to add the metals dissolved in 50 μL of water to each flask at this stage. The flasks were stoppered and maintained in the dark at ambient temperature, with maximum and minimum temperatures recorded during each series of experiments. Flasks were only briefly exposed to light when analyzed, at which time the solutions were shaken and stoppers briefly removed. Reactions were performed in triplicate. Free SO2 concentration, measured by the potassium iodate/potassium iodide-starch method, was followed over time, determinations on each set of replicates performed within 10 to 12 min. Mean time and free SO2 concentration (±SD) were then calculated and figures drawn using Excel software (Microsoft, Redmond, WA). Error bars denoting ±SD are not shown in the figures as they were smaller than the data point symbol dimensions.
Results and Discussion
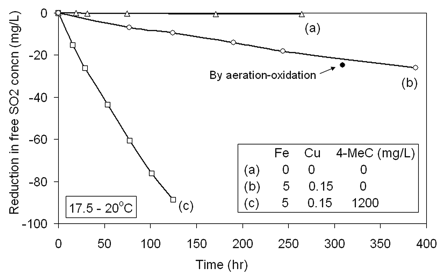
The rate of reaction of SO2 with oxygen in the wine-model solution was first examined without the addition of iron or copper. No significant reduction in free SO2 concentration was observed over the 11 days of the study (Figure 1⇓, curve a), which, as discussed above, bears out the important catalytic function of one or both of these metals. From product specifications, the concentrations of iron and copper should have been <50 and 15 μg/L, respectively, clearly insufficient to promote an observable reaction rate in these studies. The ethanol and tartaric acid were greater potential sources of metal contamination than the water.
The effect of iron and copper and 4-MeC on the reaction rate of SO2 in the wine-model system.
The average iron concentration in wines globally is given as ~5.5 mg/L, taking out some extreme values, although concentrations are generally lower when modern stainless steel equipment is used. Copper concentrations are reported to range between 0.1 and 0.3 mg/L (Ough and Amerine 1988). More recently the mean concentration of free copper ions and labile copper(II) species has been reported as 0.3 mg/L (range 0.08 to 1.39 mg/L) in a selection of white wines (Wiese and Schwedt 1997). Legal limits for “quality wine” in the European Union are 8 mg/L and 0.5 mg/L for iron and copper, respectively, and using these values for guidance, concentrations of 5 mg/L for iron and 0.15 mg/L for copper were used in the main part of this study.
Addition of these latter concentrations of the two metals to the model-wine system resulted in a steady decrease in free SO2 (Figure 1⇑, curve b). In a repeat experiment (also in triplicate) free SO2 uptake was 24.7 ± 0.2 mg/L at 309 hr (12.9 days) when measured by the aeration-oxidation method, which incidentally demonstrates that, under this condition, the iodate and aeration-oxidation methods agreed well when measuring free SO2 concentration. (Figure 1⇑). However, not all this SO2 had been oxidized to sulfate. More than one-quarter (6.9 ± 0.5 mg/L) was bound, presumably mainly to acetaldehyde formed by the oxidation of ethanol. After 20 days the free SO2 uptake was 32.2 ± 1.3 mg/L, of which 10.0 ± 0.4 mg/L was bound. Clearly, SO2 did not protect the ethanol but promoted its oxidation, which fits in with the proposed mechanism shown in scheme 2⇓, where a substantial proportion of SO2 is oxidized via route B to produce highly oxidizing SO4·− radicals. Previous estimates for mild acid conditions give the relative rates of bisulfite oxidation along routes A and B as 4:6 (Connick et al. 1995). An important conclusion is that bisulfite alone, although initially reducing Fe(III), overall does not behave as a reductant in the presence of oxygen, explaining why it does not reduce the Folin-Ciocalteu reagent (Saucier and Waterhouse 1999). Quite the reverse, SO2 enhances the oxidizing power of oxygen (Brandt and van Eldik 1995).
The effect of 4-MeC concentration on the reaction rate of SO2 in the presence of Fe (5 mg/L) and Cu (0.15 mg/L) in the wine-model system.
The relationship between 4-MeC concentration and the initial rate of SO2 reaction derived from Figure 2⇑.
The effect of Fe and Cu concentration on the reaction rate of SO2 in the presence of 4-MeC (1.2 g/L) in the wine-model system.
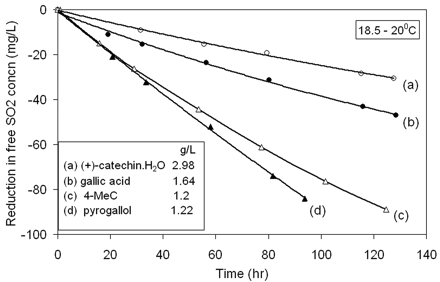
The effect of different polyphenols (9.67x 10−3 M) on the reaction rate of SO2 in the presence of Fe (5 mg/L) and Cu (0.15 mg/L) in the wine-model system.
Uptake of SO2 by a red wine (Mouton Cadet, Bordeaux) under the model-wine conditions. Mean of duplicate determinations. The average difference of any determination from the mean was 0.92% with a maximum difference of 1.96%.
Formation of sulfite radicals: radical chain initiation and redox cycling of iron.
Radical chain propagation in bisulfite oxidation. One oxygen molecule and a total of two bisulfite ions react on each turn of the cycle.
The actual importance of metals for polyphenol oxidation in wine is unclear. The iron-catalyzed oxidation of (+)-catechin has been studied in model-wine systems and reaction rate was shown to increase with increasing iron concentration (Oszmianski et al. 1996). However, significant reaction occurred without iron addition, and it was concluded that there may be an iron-independent mechanism, although that reaction might have been due to contamination by trace amounts of transition metal impurities. With regard to copper, acceleration in the degradation of (+)-catechin was demonstrated but only at a copper concentration of 0.3 mg/L and above (Clark and Scollary 2002).
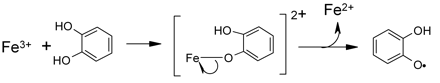
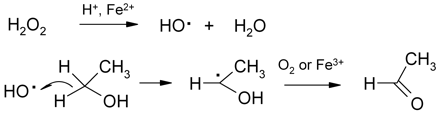
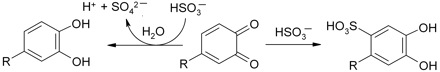
As discussed above, polyphenols also cannot react directly with oxygen in its normal triplet state. It is therefore proposed that transition metals, such as iron and copper, also play a key role in oxidative processes involving wine polyphenols (Danilewicz 2003, Waterhouse and Laurie 2006). Catechols, for instance, form complexes with metal ions such as Fe(III) and electron transfer occurs within the complex to reduce iron to the ferrous state and to oxidize the catechol to the semiquinone (inner-field electron transfer), as shown in scheme 4⇓. Semiquinones can then disproportionate or are further oxidized to produce quinones. Oxygen is consumed during this process, not by reacting with polyphenols directly but by recycling iron back to the ferric state, possibly with the assistance of copper (Cher and Davidson 1955). Oxygen is converted to hydrogen peroxide, which in the presence of iron or copper is cleaved to produce hydroxyl radicals. As mentioned above, this is a powerful oxidant (E0 = 2.7V for HO·/H2O couple), capable of removing α-hydrogen atoms from saturated hydroxy compounds such as ethanol to produce acetaldehyde (Fenton reaction, scheme 5⇓). It was proposed that an action of SO2 is to scavenge hydrogen peroxide, so preventing its destructive effect, as shown in scheme 6 ⇓(Wildenradt and Singleton 1974). This reaction is fast (McArdle and Hoffmann 1983) as is evident from its use in the determination of SO2 by the aeration-oxidation method (Buechsenstein and Ough 1978, Ough and Amerine 1988).
Possible mechanism for the reaction of SO2 with hydrogen peroxide.
Oxidation of a catechol by Fe(III).
The Fenton reaction: oxidation of ethanol.
The scavenging of hydrogen peroxide formed by the oxidation of catechols by SO2, so preventing oxidation of ethanol by the Fenton reaction.
Of the polyphenols found in wine, those containing a catechol system are the most readily oxidized, as shown in scheme 4⇑. In white wine, these include caffeic acid, (+)-catechin, and (−)-epicatechin, with two-electron formal reduction potentials E0 (2e−) = 0.79 V. In red wine there are much higher concentrations of (+)-catechin and (−)-epicatechin, together with their oligomers and polymers, collectively named the condensed tannins. Pyrogallol derivatives such as (+)-gallocatechin and (−)-epigallocatechin are also incorporated into the condensed tannins, and these derivatives are even more readily oxidized than the catechols (Danilewicz 2003). The concentration of these most readily oxidized compounds has been determined by cyclic voltammetry (Kilmartin et al. 2001, 2002) and expressed in the form of catechin equivalents (mg CE/L) (De Beer et al. 2004). For some white wines this was found to average 112 mg CE/L (range 53 to 177 mg CE/L) and for red wine 2045 mg CE/L (range 752 to 3949 mg CE/L). 4-Methylcatechol (4-MeC) was selected to represent these compounds in the model-wine system used in this study, concentrations of 45 mg/L and 823 mg/L corresponding in molar terms to the above average values in white and red wines, respectively (based on (+)-catechin monohydrate).
When 4-MeC (1.2 g/L, which corresponds to a red wine of high polyphenol content) was added to the wine-model system in the presence of iron (5.0 mg/L) and copper (0.15 mg/L), a marked increase in rate of SO2 oxidation was immediately evident (Figure 1⇑, curve c). This augmentation indicated that the catechol was oxidized at a much faster rate than the SO2, which then reacted with the hydrogen peroxide that was produced (Wildenradt and Singleton 1974). The rate of SO2 oxidation was then determined in the presence of varying concentrations of 4-MeC (Figure 2⇑). SO2 uptake at the second measurement point (between 24 and 29 hr) was recorded to calculate an approximate initial reaction rate (Figure 3⇑). Following a log-log transformation, this becomes the linear relationship y = 0.434x + 0.051 (R2 = 0.997). Clearly, the rate of reaction of SO2 is dependent on the concentration of the catechol.
Polyphenols including catechols are known to inhibit SO2 oxidation (Lim et al. 1982, Brandt and van Eldik 1995). The manner of this inhibition could occur as shown in scheme 7⇓. The sulfite radical (SO3·−) does not appear to be capable of reacting with catechols. However, its oxidation product, the peroxomonosulfate radical (SO5·−) is a much stronger oxidant (Brandt et al. 1994, Brandt and van Eldik 1995, Connick et al. 1995), the reduction potential (E3.5) for the SO5·−/HSO5− couple being 1.17 V (Das et al. 1999). The reaction shown in scheme 7⇓, where the semiquinone is formed, is thermodynamically highly favored with ΔE3.5 = 0.32 V, which translates to an equilibrium constant K = 2.6 x 105 at wine pH. As can be seen in scheme 2⇑, scavenging of this key radical would terminate chain propagation and importantly also prevent the production of the sulfate radical (SO4·−) along route B, thus protecting ethanol from oxidation. It seems likely therefore that in wine, and particularly red wine, the interaction of SO2 with oxygen as depicted in scheme 2⇑ should be effectively blocked by polyphenols.
Scavenging of peroxomonosulfate radicals by catechols so preventing radical chain propagation, which results in inhibition of SO2 autoxidation.
To examine this inhibitory effect, the experiment in which 50 mg/L 4-MeC had been added to the model system was repeated and followed for 12 days, by which time 51.3 ± 1 mg/L free SO2 had been consumed as measured by the iodate method (Figure 2⇑). Using the aeration-oxidation procedure, free SO2 was reduced by 49.1 ± 1.7 mg/L, which again shows good agreement between the two assay methods. However, bound SO2 was then increased by only 1.1 ± 1.2 mg/L, in marked contrast to the results obtained when SO2 was oxidized in the absence of the catechol, when ~30% of the free SO2 taken up was not oxidized but was bound. Evidently, even these low concentrations of catechol, which are intended to simulate white wine conditions, were sufficient to inhibit SO2 autoxidation and so protect the ethanol from SO2-oxygen derived oxidizing radicals. In this experiment, ~2 mole equivalents of SO2 were consumed relative to the initial catechol concentration, indicating that the SO2 reacts not only with hydrogen peroxide but also with the quinone; the mechanism of this reaction is currently being investigated (see discussion below). To provide further evidence of the inhibitory action of catechols on SO2 oxidation, the remaining solutions from a second triplicate experiment, in which the effect of 350 mg/L 4-MeC on SO2 oxidation was studied, were combined after 6 days. Free SO2 was reduced by 64.5 mg/L, but when the free SO2 concentration returned to its initial value (92.5 mg/L), bound SO2 had increased by <1 mg/L (mean of duplicate determinations). Clearly, there had been little ethanol oxidation. These results demonstrate not only that SO2 autoxidation is unlikely to occur in wine as a result of the radical scavenging action of polyphenols but also that in this system SO2, by reacting with hydrogen peroxide, prevented ethanol oxidation by way of the Fenton reaction.
The importance of iron and copper in the oxidation of 4-MeC (1.2 g/L) was then examined. When the catechol was exposed to air in the presence of SO2, iron (5 mg/L), and copper (0.15 mg/L) in the model solution, there was a relatively rapid fall in SO2 concentration because of the production of hydrogen peroxide (Figure 1⇑, curve c). In contrast, when neither metal was added, essentially no reaction was observed over 5 days (Figure 4⇑, curve a). The reduction in free SO2 was only 0.5 ± 0.3 mg/L, which was at the limit of accuracy of the experimental method. Catechol oxidation was reduced to an extremely slow rate, and it is proposed that any remaining oxidation that might be observed in the longer term would most likely be due to trace metal impurities. These results gave clear evidence as to the crucial importance of one or both of these metals in polyphenol oxidation.
When iron (5 mg/L) was added, a surprisingly small augmentation in reaction rate was observed (Figure 4⇑, curve c) compared with that when 0.15 mg/L of copper was also present (curve f ). Raising iron concentration to 10 mg/L only produced a small additional rate increase (curve d), while copper (0.15 mg/L) alone also only produced a small increase in reaction rate (curve b). The two metals together produced a far greater rate increase than the sum of rates observed when they were tested singly. This synergism was evident at very low copper concentrations (curve e). Thus, the rate of catechol oxidation is markedly influenced by copper concentration provided iron is also present (compare curves b, e, f, and g). The ability of iron to catalyze the oxidation of catechols is dependent on redox cycling, as ferrous ions, which are produced when ferric ions are reduced by catechols, must be reinstated to the ferric state to continue the catalytic process. A possibility is that copper facilitates this redox cycling, which may be rate-limiting by providing a more effective interaction with oxygen, possibly by forming a CuO2+ species (Balla et al. 1992). The overall process may then be represented as in scheme 8⇓, in which catechols are oxidized both by Fe(III) and hydroperoxyl radicals, the whole process driven by the redox cycling of the metals.
Redox cycling of copper and iron while catalyzing the oxidation of catechols to produce quinones and hydrogen peroxide.
A consideration is the fate of the quinone that is produced when the catechol is fully oxidized, as it may also contribute to the uptake of SO2 in this model system. Studies of the interaction of bisulfite with 1,4-benzoquinone (LuValle 1952, Youngblood 1986) have shown that at pH 3.5 approximately half was reduced back to 1,4-benzenediol and half underwent Michael-type 1,4-addition to the sulfonic acid. The mechanism is very pH sensitive in this pH region, as at 4.5 only the sulfonic acid was observed. These transformations can be represented as shown in scheme 9⇓ when applied to a catechol. Saucier and Waterhouse (1999) proposed the reduction mechanism to explain the apparent synergism between SO2 and (+)-catechin in the Folin-Ciocalteu assay. The addition route is an alternative possibility, with the irreversible formation of the stable sulfonic acid, as 3,4-dihydroxyben-zenesulfonic acid is produced when catechol is oxidized by tyrosinase in the presence of SO2 at pH 6.5 (Wedzicha et al. 1987). Quinones are strongly electrophilic and are known to undergo addition reactions with nucleophiles such as thiols (Cilliers and Singleton 1990). Evidently, bisulfite should be capable of reacting with quinones in wine conditions. The manner of this interaction may explain how SO2 prevents the loss of odorant thiols on oxidation of wine and wine-model systems, which otherwise would have reacted with quinones (Blanchard et al. 2004). It may also explain how it could prevent wine browning that may be due to quinone and semiquinone-derived reaction products. However, it is possible that the interaction of SO2 with quinones derived from flavanols and polymeric derivatives (condensed tannins) may be different to that of 4-MeC because of their much greater structural complexity and possibility of competing reactions.
Possible reactions of bisulfite ions with quinones.
The reaction of SO2 was also examined in the presence of equimolar concentrations of different polyphenols (Figure 5⇑). The rate of reaction decreased in the order pyrogallol (E0 = 0.71 V) > 4-MeC (E0 = 0.75 V) > gallic acid (E0 = 0.80 V), in line with the increasing two-electron formal reduction potentials of their respective quinone-polyphenol couples and hence supposed susceptibility to oxidation (Danilewicz 2003). Most unexpected, however, was the slowness of the reaction in the presence of (+)-catechin. The formal reduction potential of its redox couple and hence its rate of oxidation should be very similar to that of 4-MeC. Consequently, the 1H-NMR spectrum of the (+)-catechin sample was checked. It proved entirely consistent with its structure and was identical to that published without any visible impurities (Aldrich Library 1993). This anomalous behavior is unexplained. However, this order of reactivity was also found by Wildenradt and Singleton (1974). Acetaldehyde was produced more slowly when (+)-catechin was oxidized than gallic acid, and both showed slower rates of acetaldehyde production than 4-MeC.
The rate at which SO2 concentration is reduced in a real wine on exposure to oxygen was also examined to see how it compared with the model system. A difference is the presence of much larger amounts of substances that bind bisulfite reversibly, such as carbonyl compounds. The free SO2 concentration of the wine was raised to 84.4 mg/L, which resulted in a bound concentration of 70.8 mg/L, with at least 6 hr required to achieve equilibrium. Exposure to air in the same manner as that of the model experiments gave a prompt linear reduction in total SO2. As free SO2 concentration progressively declined, bound SO2 dissociated and was drawn into reaction (Figure 6⇑). The rate of reaction was about the same as observed in the model-wine system containing 1.64 g/L of gallic acid (Figure 5⇑).
Conclusion
These studies have provided evidence as to the catalytic importance of iron and copper in the autoxidation not only of polyphenols but also of SO2 in a wine-model system. Furthermore, they indicate that nonmetal catalyzed pathways, if they do exist, are so much slower that they are unlikely to play a significant part in oxidative processes in wine. SO2 autoxidation in the model solution results in an increase in bound SO2, presumably due to the oxidation of ethanol to acetaldehyde, which is prevented by 4-MeC. This observation supports the proposal that SO2 autoxidation is a radical chain reaction in which powerful intermediate oxidizing radicals are produced. These are intercepted by radical scavenging polyphenols, which break the radical chain process and prevent the interaction of SO2 and oxygen in wine conditions. It is therefore concluded that SO2 exerts its antioxidant action primarily by reacting with hydrogen peroxide.
The simple concept that catechols and SO2 are individually antioxidants should be reconsidered. When catechols are exposed to oxygen, hydrogen peroxide is produced, which, by way of the Fenton reaction, will oxidize ethanol. Clearly, catechols provide a route by which oxidation can occur, and in that respect promote oxidation, and this also applies to ascorbic acid. In the presence of oxygen, SO2 also promotes oxidation, which is prevented by the radical scavenging action of polyphenols. Overall, therefore, SO2 is required to scavenge the hydrogen peroxide and polyphenols are required to block the SO2-oxygen interaction, and it is only then, when polyphenols and SO2 are combined, that the full antioxidant action is revealed in wine.
These results may prompt those studying oxidative processes in model systems to consider the inclusion of appropriate levels of both iron and copper. The possible involvement of manganese is yet to be examined. The results indicate that the rate of SO2 oxidation should be dependent on the concentration of the more reducible polyphenols and is also likely to be highly sensitive to copper concentrations since iron will invariably be present. Perhaps copper should not be considered merely as a potential cause of wine instability with respect to copper casse.
- Received March 2006.
- Revision received July 2006.
- Copyright © 2007 by the American Society for Enology and Viticulture